Genesis® Malleable Penile Implant
The Power of Simplicity

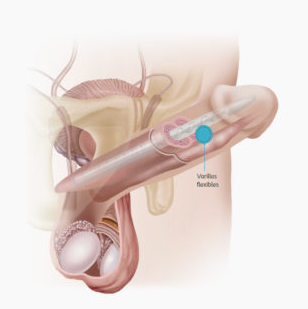
The Genesis® malleable implant offers a solution for patients with finger or hand dexterity issues, hand muscle fatigue or limited reach and range of mobility. It is coated with HydroVantage® hydrophilic coating facilitating easy placement and surgeon choice of aqueous dipping solution in the operating room.
The Genesis malleable penile implant consists of two firm but flexible rods that are placed into the shaft of the penis. There are no other parts to this implant. To have an erection, you simply hold the penis and move it into the desired position. When you are finished, you bend it back down to conceal it.
Other features include:
- No cables or moving internal parts to compromise reliability
- Trimmable – custom sizing to fit to each individual’s corporal length
- Easy assembly – no special tools required, simply connect
- Multiple sizes – 9.5 mm, 11 mm, 13mm diameter
Learn about Coloplast solutions
Genesis Malleable Penile Prosthesis Brief Statement
Intended Purpose
The Genesis Malleable Penile Prosthesis is a flexible silicone elastomer device designed to be implanted into the penis for the management of erectile dysfunction (commonly known as impotence).
Indications
The Genesis Malleable Penile Prosthesis (Prosthesis) is designed for the management of erectile dysfunction (impotence) stemming from a variety of causes, including: epispadias, pelvic fracture, spinal cord injury or disease, prostatectomy, cystectomy, abdominal-perineal resection, multiple sclerosis, diabetes mellitus, alcoholism, arteriosclerosis and hypertensive vascular disease, priapism, and Peyronie’s disease. The Prosthesis may also be used in selected patients with psychogenic impotence.
Contraindications
The Genesis Malleable Penile Prosthesis is contraindicated in patients who have one or more of the following conditions: patients with an active infection present anywhere in the body, especially urinary tract or genital infection, patients with a documented hypersensitivity or allergic reaction to silicone, patients with unresolved urinary problems such as an elevated residual urine volume secondary to bladder outlet obstruction or neurogenic bladder.
Warnings
It is the responsibility of the physician to advise the prospective patient prior to surgery, of the warnings and precautions associated with the use of this product and the associated surgical risks. The Genesis Malleable Penile Prosthesis should only be implanted by physicians experienced in the surgical procedures and techniques involving implantation of a malleable penile Prosthesis. The physician should maintain adequate education to conduct this procedure. Implantation of a penile Prosthesis may make natural erections difficult to achieve. It may make some other interventional treatment options not possible.
Implantation of a penile Prosthesis may result in penile shortening, curvature or scarring. Pre-existing penile scarring or contracture may make surgical implantation more complicated or impractical. Failure to evaluate and promptly treat erosion may result in infection and loss of tissue. Men with diabetes as well as immunocompromised patients, may have an increased risk of infection which could result in permanent damage to tissue/organs. The Prosthesis should be used with caution in patients with borderline bladder decompensation, an indwelling catheter, or enlargement of the prostate.
Precautions
A thorough preoperative consultation should include a discussion between the patient and physician of other available treatment options and their risks and benefits. Patients should be informed that erections achieved with a malleable penile Prosthesis may differ from original erection (e.g. not of equal length or girth) compared to what was previously experienced with natural erections. Removal of an implanted Prosthesis without timely reimplantation of a new Prosthesis may make subsequent reimplantation more difficult.
Adverse Reactions
Adverse events are known to occur with penile protheses procedures and implants; some may require revision surgery or removal of the implant. Adverse events following penile protheses implantation may be de novo, persistent, worsening, transient, or permanent. Adverse Reactions include: deformity, delayed/Impaired/Abnormal wound healing, erosion/extrusion, fistula, foreign body reaction, hematoma/Seroma, hemorrhage/bleeding, hypersensitivity/allergic reaction, infection (local or systemic), inflammation (i.e., edema, erythema), irritation, migration, necrosis, pain/discomfort, paraphimosis/acquired phimosis, perforation or injury of soft tissue (e.g., muscles, nerves, vessels), penile structures, or organs (e.g., urethra), scar tissue, sexual dysfunction, tactile disorders (e.g., hypoesthesia, numbness), urethral obstruction/occlusion, urinary tract infection, and voiding symptoms.
The occurrence of these events may require one or more subsequent surgeries which may or may not always fully correct the complication.
The information provided is not comprehensive with regard to product risks. For a comprehensive listing of indications, contraindications, warnings, precautions, and adverse events refer to the product’s Instructions for Use. Alternatively, you may contact a Coloplast representative at 1-800-258-3476 and/or visit the company website at www.coloplast.com.
Caution: Federal law (USA) restricts this device to sale by or on the order of a physician.
PM-16954 05.2021
References
- Pescatori ES, Goldstein I. Intraluminal device pressures in 3-piece inflatable penile prostheses: the pathophysiology of mechanical malfunction. J Urol. 1993 Feb;149(2):295-300.
- Scovell JM, Ge L, Barrera EV, Wilson SK, Carrion RE, Hakky TS. Longitudinal and Horizontal Load Testing of Inflatable Penile Implant Cylinders of Two Manufacturers: An Ex Vivo Demonstration of Inflated Rigidity. J Sex Med. 2016 Nov;13(11):1750-1757.
- Pritchard, Charles, MD, et al. “Comparison of AMS 700 CX and Coloplast Titan Inflatable Penile Prosthesis Cylinders Subjected to In-Vitro Cyclic Buckling.” Sexual Medicine Society of North America Fall Meeting, Poster 111, 2008.
- Garber BB. Mentor Alpha 1 inflatable penile prosthesis: patient satisfaction and device reliability. Urology. 1994 Feb;43(2):214-7.
PM-29359
